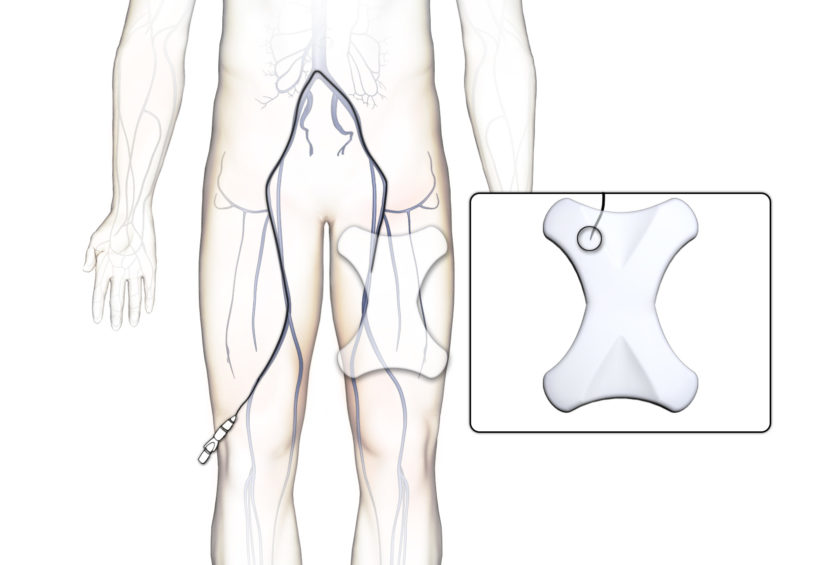
SAGE’s near-field low-frequency technology both meets the need for a cost-effective, bedside method of tracking inserted devices during medical procedures. SAGE technology is essentially immune to interference from nearby metal objects and magnetic sources, and has a 3D location and orientation accuracy sufficient for many procedures. Please contact Lucent Medical to see if SAGE technology is right for your particular medical device.
Our Services
Electrical and Mechanical Engineering
We provide the EE and ME necessary to create a sensor module for your medical application, and clinical, and patient environment. During the design process, we provide circuit design, board layout, 3D renderings, CAD drawings, and several levels of prototypes. Once a design is approved, we will build engineering evaluation and validation prototypes for safety testing and agency approvals.
Disposables Design
We can provide mechanical design services to customize your disposable medical device for SAGE tracking. The SAGE coil design will be tailored to match the requirements of the tracked device and medical procedure.
Firmware and Application Software
Firmware and application software will be written and customized for your application.
Testing and Validation
We can perform required testing and validation, manage agency approvals, and provide final documentation for submission to the FDA for marketing approval.
Finished Product Manufacturing and Delivery
We can provide production ramp up and ongoing contract manufacturing support, to deliver fully finished products to your distribution center.
SAGE
- Customized. Lucent’s SAGE tracking technology can be adapted to your particular device.
- Front to back engineering. We help develop your product from idea, to prototyping, to contract manufacturing.
- Applications. SAGE technology can be applied to catheters, stylets, guidewires, probes, needles, and other devices to which a small wire coil can be attached.
- Ease-of-use. The SAGE sensor module uses a standard USB interface to connect to its display system.
- Interference Free. No effect on tracking from external metal objects or magnetic sources.
- Accuracy. 3D Location and orientation accuracy is adequate for many medical procedures.
Contact Us
To find out more about SAGE technology, please fill out and submit the form below, or give us a call at 425-822-3310, extension 28.

